Valence electrons are electrons in the outer most shell or orbit and there are 4 valence electrons present in the lead (Pb) outermost shell.
What is the valency of Pb in PbO₂?
PbO2 is an oxide where the oxidation state of lead is +4 with chemical name Lead (IV) oxide.
Does Pb show variable valency?
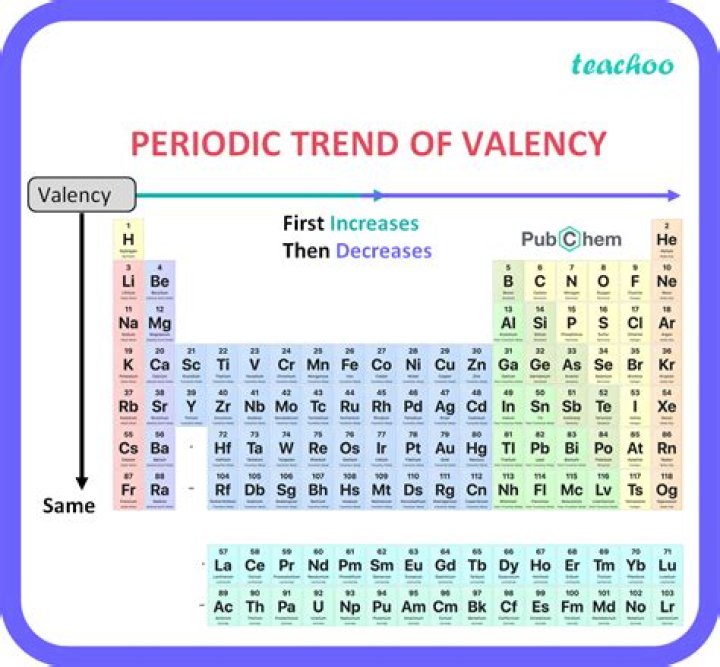
Lead (Pb) and tin(Sn) show variable valencies of +2 and +4.
What is the valency of Pb in PbCl4?
Originally Answered: What is the exact valency of lead? Due to inert pair effect, Pb shows +2 valency.
What is the oxidation number of Pb in Pb3O4?
The real chemical formula of Pb3O4 is PbO2 x 2PbO. It means that one atom of lead has oxidation number +4 and the other two atoms have +2.
What is the valence electrons for potassium?
Atomic number of potassium atom is 19. Its electronic configuration is 2,8,8,1 so, its valency is 1.
How do you find variable valency?
The steps to calculate the valency are:
Write the electronic configuration of an element.If the outermost shell has less than or equal to four electrons then the valency is the number of outermost electrons.If the outermost shell has more than four electrons then the valency is 8-(the number of outermost electrons).
What is the valency of lead Pb in lead nitrate?
Valency of nitrate is 2. But chemical formula for lead nitrate is Pb(NO3)2 how ?? It should be PbNO3.
What is the name of Pb2+?
Lead (II) ion. Lead(2+) ion. Pb2+