Water has three normal modes of vibration, all of which are IR active. Carbon dioxide, a linear molecule, has 4 normal modes of vibration. Even though it does not have a permanent dipole moment, the dipole moment changes during 3 of the 4 modes, so carbon dioxide can absorb in the IR.
Which vibrational modes of H2O are IR active?
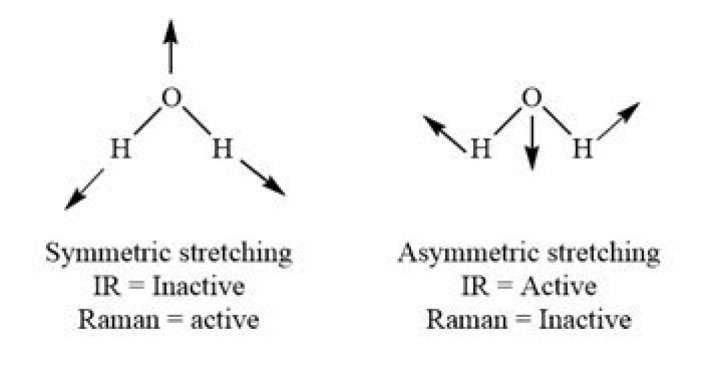
The IR spectrum of H2O does indeed have three bands as predicted by Group Theory. The two symmetric stretches v1 and v2 occur at 3756 and 3657 cm-1 whereas the bending v3 motion occurs at 1595 cm-1. H2O has two stretching vibrations as well as one bending vibration.
Is hydrogen IR active?
It is known that symmetrical diatomic molecules like nitrogen, oxygen and hydrogen, do not absorb infrared radiation, even though their vibrational frequencies are in the infrared region.
What makes something IR active?
Some bonds absorb infrared light more strongly than others, and some bonds do not absorb at all. In order for a vibrational mode to absorb infrared light, it must result in a periodic change in the dipole moment of the molecule. Such vibrations are said to be infrared active.
What is IR active and IR inactive?
For highly symmetric polyatomic molecules possessing a center of inversion, the bands are IR active (Raman inactive) for asymmetric vibrations to i and for symmetric vibrations to i the bands are Raman active (IR inactive). A mode can be IR active, Raman inactive and vice-versa however not at the same time.
Which of the following molecules is not IR active?
Homonuclear diatomic molecules do not show IR spectra. Homonuclear diatomic molecules such as H2, N2, O2, do not have a permanent dipole moment, nor the stretching of the atoms about the bond gives rise to any dipole moment. Hence homonuclear molecules do not show any IR spectra.
Is H2O2 IR active?
Absorption of infrared radiation by H2O2 is relatively weak. For reference, the principal absorption bands for water are: 0.85, 0.98, 1.18, 1.46, 1.98, 2.97, and 6.1 m.
Is n2o IR active?
Characteristic Vibrations of N2O (C v symmetry)
All three of the modes shown here are infrared-active to some degree, making them capable of absorbing and scattering infrared heat radiation from the Earth before it can escape to space.
Are linear molecules IR active?
Since the linear molecule is centrosymmetric, the Raman band does not appear in the IR, nor do any of the IR bands appear in the Raman spectrum. Hence, for a linear AB2 molecule we would expect 2 IR active bands and 1 Raman band.
Why is HCl IR active?
If we stretched the bond so far as to break the bond and produce the two original neutral atoms, there would be no dipole moment. Therefore, as we lengthen the bond in HCl, the dipole moment gets smaller. Because the dipole moment of HCl changes during a stretching vibration, it absorbs infrared radiation.
Is symmetric stretching IR active?
The symmetric stretch does not change the dipole moment so it is not IR active.
Is carbon tetrachloride IR active?
CCl4 and CS2 are having dipole moment zero or no dipole. But both are having characteristic vibrational bands in the mid IR range.
Is ozone IR active?
The IR spectrum of Ozone (O3 ) exhibits three IR active fundamental vibrational bands at 1355, 1033, and 528 cm−1 .
Why is CO2 IR inactive?
Since carbon dioxide is linear it has 3n−5=4 vibrations and they are pictured below. The symmetric stretch does not result in a change (of the initially zero dipole moment), so it is ir-inactive. The asymmetric stretch does result in a change in dipole moment so it is ir-active.
Is H2 Raman active?
For this reason, homonuclear and thus nonpolar molecules such as H2 or N2 are Raman-active. Their rotational and vibrational spectra can be measured using the Raman effect, although they are not accessible to microwave or infra-red spectroscopies because the transitions are forbidden by symmetry.
Is ethene IR active?
The IR spectrum of ethene is rather uncomplicated due to its simple structure. It has a strong band at 900 cm-1 (11 µm) due to H-C-H out- of- plane wagging.